Marleen Hoefnagel
Visit: 5 months

Electrochemical reactions play a key role in the transition from fossil fuels to renewable energy, and from a polluting and cradle-to-grave approach to sustainable and circular alternatives.
For this transition active, selective and stable catalysts are needed. Homogeneous catalysts often have high activity and selectivity and allow for studying structure-activity correlations. Solubility and reusability of these homogeneous catalysts pose a big challenge which may be overcome by immobilizing them on electrodes.
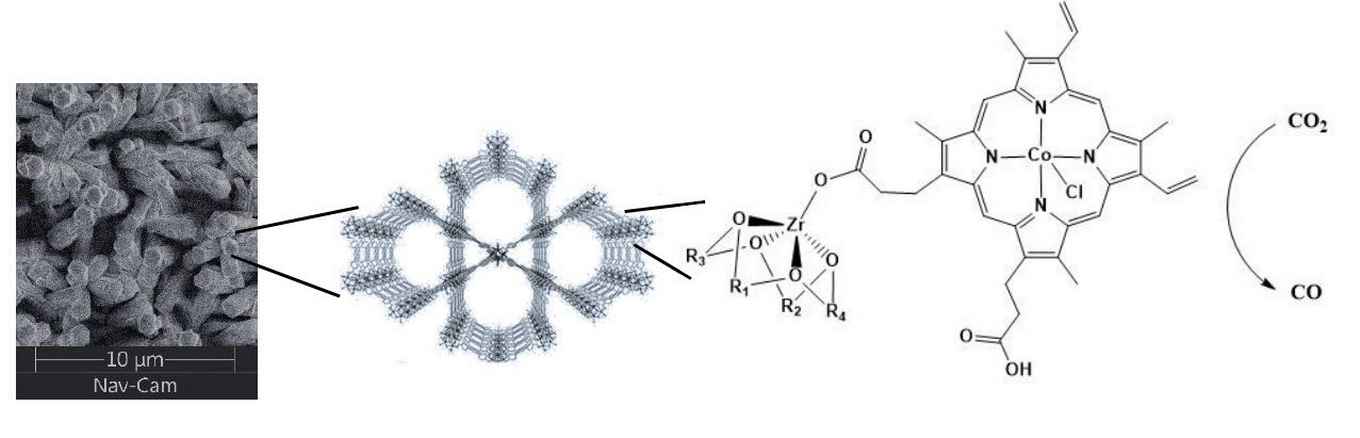
Metal Organic Frameworks (MOFs) are highly symmetrical, porous structures that consist of metal nodes and organic linkers. MOFs are very interesting scaffolds for robust immobilization of homogeneous catalysts as they bind the catalyst covalently and offer an extremely high surface area for potential catalytic sites when these MOFs are attached to electrodes.
We can currently form MOFs that are catalytically active and stable over prolonged electrolysis experiments, but we can only activate a small portion of catalytic sites due to e- transfer problems. We are currently trying to understand the underlying principles that govern the efficiency of the CO2/O2 reduction reactions towards specific products and the design rules that allow us to address a high percentage of catalytic sites within the MOFs.
We anticipate a significant role for the unique mass- and charge transport in MOF pores and believe this is the key to unlocking the full potential of MOF electrocatalysts.
During my stay in Uppsala I will work on synthesizing a range of MOFs with a specific focus on gaining control over mass- and electron transport within the framework during catalysis. Via this approach, we hope to improve the conductivity and catalytic performance of or MOFs towards the electrocatalytic CO2 and O2 reduction reactions.