Dieuwertje Modder wins poster competition at national student symposium
26 March 2019
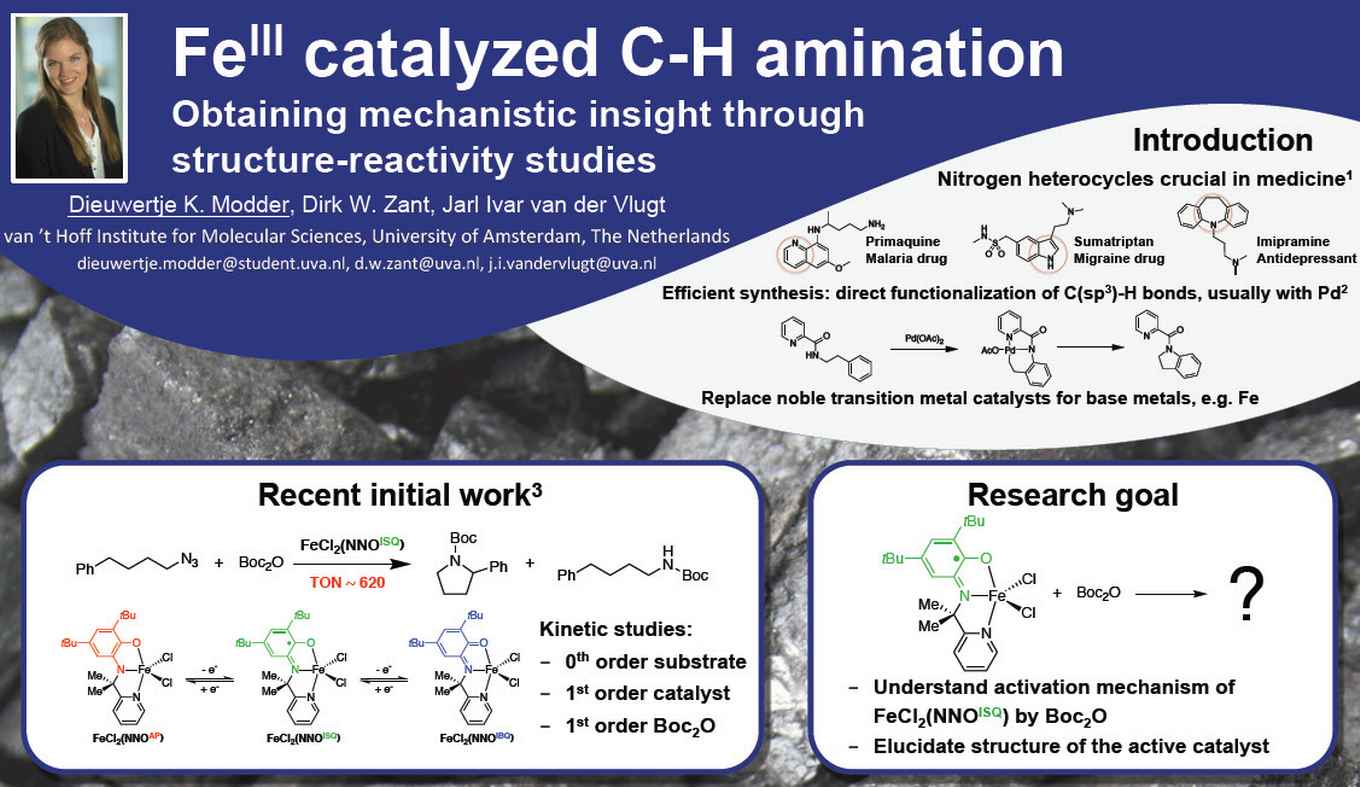
In her research project, Modder contributes to the understanding the activation mechanism of iron(III) catalysed C-H amination for the synthesis of pharmaceutical building blocks. It is supervised by PhD student Dirk Zant and research leader Dr Jarl Ivar van der Vlugt. Van der Vlugt investigates the use of redox-active ligands in catalysis, for example as electron reservoirs to enable catalytic cycles involving two electrons for base metals (which typically undergo cycles involving just electron). In earlier research an iron(III) catalyst containing such a redox-active ligand was developed, displaying extreme activity in the intramolecular C-H amination reaction. This reaction is important in the synthesis of heterocyclic molecules containing a nitrogen atom, which is relevant for the production of pharmaceuticals. It is typically catalyzed by noble metals such as palladium, so the discovery of a cheaper and more abundant base metal catalyst is of great relevance.
Structure-reactivity study
Interestingly, kinetic studies indicate that the synthesized iron(III) complex is not the actually active catalytic species. It is first activated by one of the substrates, which is the rate determining step. Dieuwertje Modder hopes to shed light on this by synthesizing the same iron(III) complex but with slight variations, and determine how these changes influence the activity of the catalyst. Her structure-reactivity studies will hopefully provide insight into the essential structural elements of the catalyst, and indicate how the complex is activated. Based on this information, improvements can be made to facilitate the activation and as a result increase the reaction rate.
